It is the 10th week of a Phase III oncology trial. Site 19 has screened 24 patients. Only 2 reached randomization. The remaining patients dropped off between identification and enrollment.
The site coordinator is juggling five different logins, each with its own password policy, each demanding another reset. Meanwhile, a patient sits at home staring at yet another “Reset Your Password” email. It’s the third one today. The link doesn’t work. Again. They close the laptop. They’re done.
One patient stopped trying to enroll. The password reset email failed three times. The process did not recover.
The sponsor has $50 million riding on this study while the money is getting hemorraged through broken workflows, fragmented systems, and one Excel spreadsheet after another.
And with that click, another potential participant disappears from the trial.
The Common Thread Between Patient Identification, Engagement, and Retention: Why Clinical Recruitment Succeeds or Fails at the Platform Layer
For decades, the clinical research industry has treated patient recruitment a marketing problem to be solved with higher ad spends, flashy outreach campaigns, and an ever-expanding "toolkit" of point solutions. Yet despite the proliferation of patient recruitment strategies, the numbers haven’t budged. Nearly 80% of clinical trials still fail to hit clinical trial enrolment timelines, and a staggering 30% of patients drop out mid-study.
If the problem were simply a lack of willing participants, these billions of dollars in investment would have yielded better results. The reality is more systemic: recruitment fails because of a profound breakdown in architecture.
For Sponsors, CROs, and Clinical Operations heads, the takeaway is clear: identification, engagement, and retention are not three isolated phases of a trial. They are interconnected pulse points of a single data lifecycle. When that lifecycle is fragmented across closed EHRs, siloed site files, and a dozen different patient apps, patient dropout rates climb and clinical trial site management becomes unworkable and breaks.
To fix the pipeline, leadership must stop looking at operational band-aids and start looking at the platform layer for healthcare engagement.
The Architectural Imperative: Moving Beyond Siloed Solutions
In the process of rushing to digitize, the healthcare industry has created "Frankenstein’s monster" in clinical tech. A sponsor might procure a best-of-breed data-mining tool for identification, a separate eConsent module for enrollment, and a standalone ePRO app for retention. The growth of decentralized clinical trials has only compounded this problem, adding remote patient monitoring tools and telehealth layers on top of an already fragmented eClinical platform stack.
On paper, these tools work. In practice, they create an unsustainable integration tax on sites and a disjointed, exhausting experience for the patient. A true digital platform for patient engagement is not a gallery of loosely connected apps; it is a unified foundation that harmonizes data streams and provides a single, longitudinal view of the patient from the first "click" to the final study visit.
When clinical operations are anchored to a robust platform layer, the friction between identifying a candidate and actually keeping them enrolled begins to vanish.
Patient Identification & Data Integration
Traditional patient identification is often a "look back" exercise: manual chart reviews and queries of yesterday's data. This is inherently reactive, relying on static snapshots of health that rarely reflect the real-time clinical reality of a patient.
When identification is pushed to the platform layer, it becomes an active, living process. By leveraging deep clinical trial integration with health system infrastructure, a modern platform can ingest and normalize real-world data (RWD) and EHR feeds; enabling AI-powered patient identification that flags eligible candidates the moment they interact with the healthcare system. This transforms real-world data in clinical trials from a retrospective tool into a live recruitment engine.
The "secret sauce" here is study setup integration. During the design phase, the platform must be configured to map complex protocol criteria directly to standardized data elements (like FHIR resources). When this integration is foundational rather than an afterthought, clinical ops teams can stop guessing at feasibility and start alerting physicians to eligible patients while they are still in the exam room.
Engagement & Workflow Orchestration
Identification is only the spark; digital patient engagement is the engine. Far too often, "engagement" is used as a fancy word for a passive portal where patients are expected to find their own answers. This "pull" strategy rarely works in high-stakes clinical environments.
A strategically designed platform layer turns engagement into an automated, proactive workflow. It bridges the gap between the busy investigator and the overwhelmed patient by orchestrating the right message at the right moment. Whether it’s an automated SMS triggered by a specific lab value, a video explaining a complex procedure, or a frictionless eConsent flow, the platform handles the choreography behind the scenes.
By embedding these workflows directly into the tools site coordinators already use, the platform eliminates the "swivel chair" effect. Sites don’t have to toggle between five screens to talk to one patient, which significantly reduces screen failure rates and the drop-off that happens between screening and enrollment—directly improving the overall patient enrollment rate.
Retention & Experience Design
Patient retention is essentially a measure of how much "friction" we ask a participant to endure. If a trial requires a patient to manage three different logins, carry a secondary device, and manually type in data that their smartphone is already tracking, they will eventually quit.
When driven by a centralized platform layer for healthcare engagement, that friction is replaced by a unified "digital front door." A single interface allows patients to manage their schedule, handle travel logistics, and complete surveys without ever seeing the messy plumbing of the underlying systems.
Furthermore, a platform-driven approach allows for predictive retention. By analyzing engagement telemetry—like a patient taking longer to complete a patient-reported outcomes (PRO) diary or missing a remote patient monitoring check-in—the system can flag a "risk of dropout" to the site team before it actually happens. It moves the site from being reactive to being proactive.
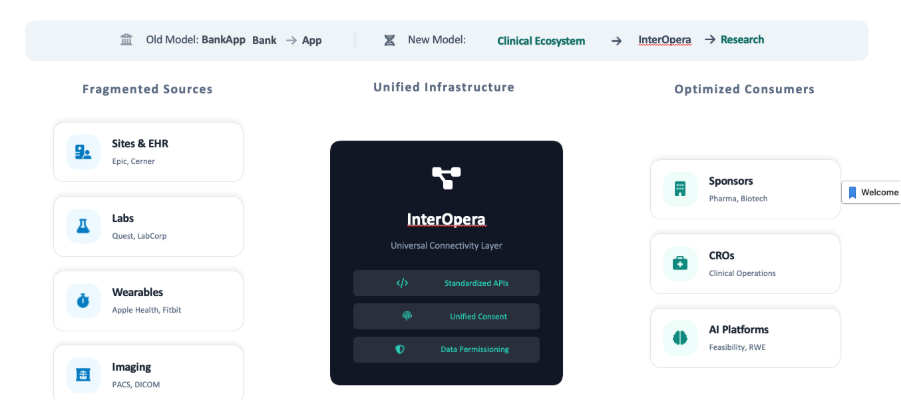
Interoperability & Platform Architecture
The "connective tissue" that holds this all together is clinical research interoperability. Without the ability to move data securely across organizational boundaries, even the best-looking platform is just another silo.
Building an interoperable clinical research ecosystem requires moving beyond "plug-ins" and toward an API-first architecture. This means utilizing modern standards like HL7 FHIR and CDISC to ensure the platform speaks the same language as the hospital’s EHR.
A sophisticated architecture doesn't try to replace every legacy system. Alternatively, it acts as an intelligent orchestration layer. It bi-directionally syncs data, ensuring that a medication change in a patient’s primary care record is instantly reflected in the trial's EDC system. This is the only way to truly dissolve the wall between clinical care and clinical research.
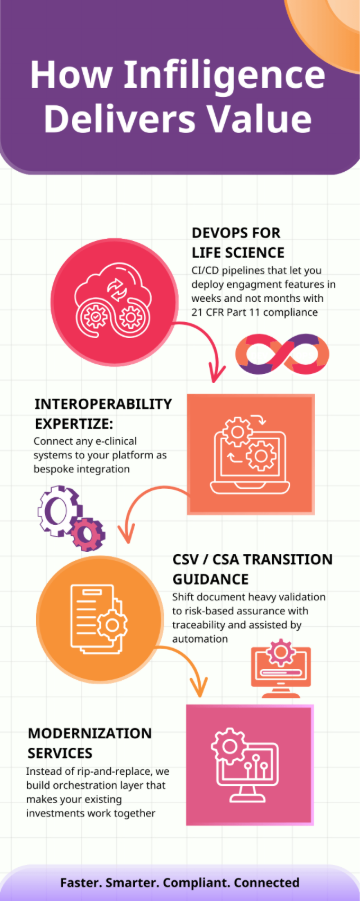
Compliance, CSV, and the Move Toward CSA
The biggest roadblock to this vision is often the perceived regulatory burden. Historically, the industry has been paralyzed by computer system validation (CSV)—a document-heavy, rigid process that makes even the smallest software update feel like a mountain-climbing expedition.
However, the FDA is signaling a major shift toward computer system assurance (CSA). This represents a paradigm shift for digital transformation leaders.
Where CSV was about "testing everything" to produce a paper trail, CSA is about "testing what matters." It encourages a risk-based monitoring approach aligned with GCP compliance and 21 CFR Part 11 requirements, focusing effort on features that impact patient safety and data integrity while using automated testing for lower-risk integrations. This allows platform teams to be agile—deploying new engagement features and fixing UX bottlenecks in weeks rather than months. A compliant platform is no longer a static relic; it is a secure, evolving ecosystem.
The Future: Interoperable, Platform-Driven Clinical Research
The line between a successful trial and a failed one is no longer just the quality of the molecule; it’s the quality of the architecture. Sponsors and CROs who continue to treat recruitment as a series of disconnected operational tasks will continue to pay the "silo tax" in the form of delayed timelines and high dropout rates.
The future belongs to the digital platform for patient engagement. By anchoring trial operations to an interoperable platform—one that connects the patient's daily life with the researcher's clinical workflow—the industry can finally provide a trial experience that feels like it belongs in the 21st century.
When the platform layer is built correctly, identification leads naturally to engagement, and engagement secures retention. Patient-centric clinical trials stop being an aspiration and become the operational default. Only then can clinical trial technology move research at the speed that patients deserve.
.png)

